Types of radiation
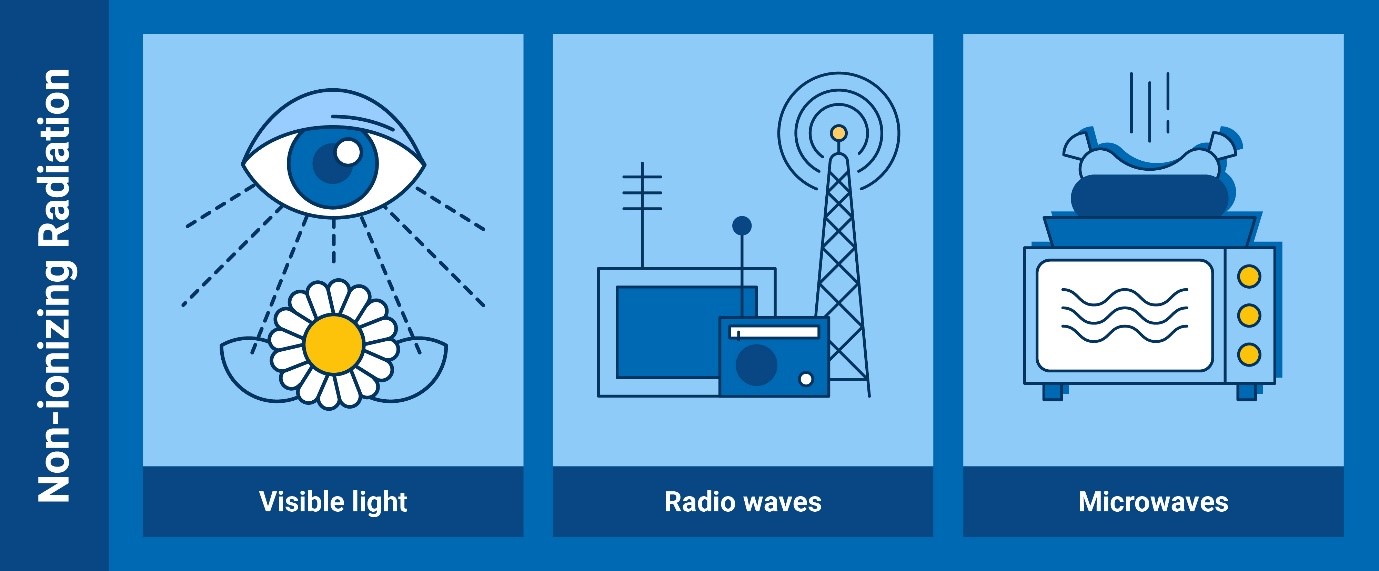
Non-ionizing radiation
Some examples of non-ionizing radiation are the visible light, the radio
waves, and the microwaves (Infographic: Adriana Vargas/IAEA)
Non-ionizing radiation is lower energy radiation that is not energetic enough
to detach electrons from atoms or molecules, whether in matter or living
organisms. However, its energy can make those molecules vibrate and so
produce heat. This is, for instance, how microwave ovens work.
For most people, non-ionizing radiation does not pose a risk to their health.
However, workers that are in regular contact with some sources of
non-ionizing radiation may need special measures to protect themselves from,
for example, the heat produced.
Some other examples of non-ionizing radiation include the radio waves and
visible light. The visible light is a type of non-ionizing radiation that
the human eye can perceive. And the radio waves are a type of non-ionizing
radiation that is invisible to our eyes and other senses, but that can be
decoded by traditional radios.
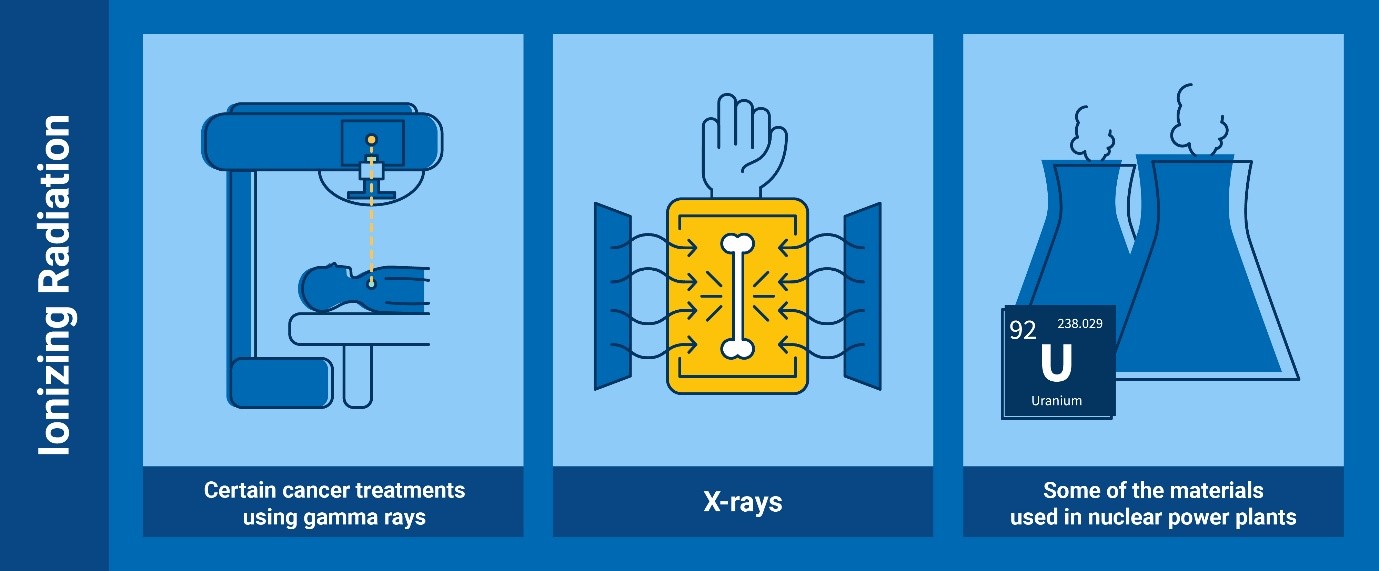
Ionizing radiation
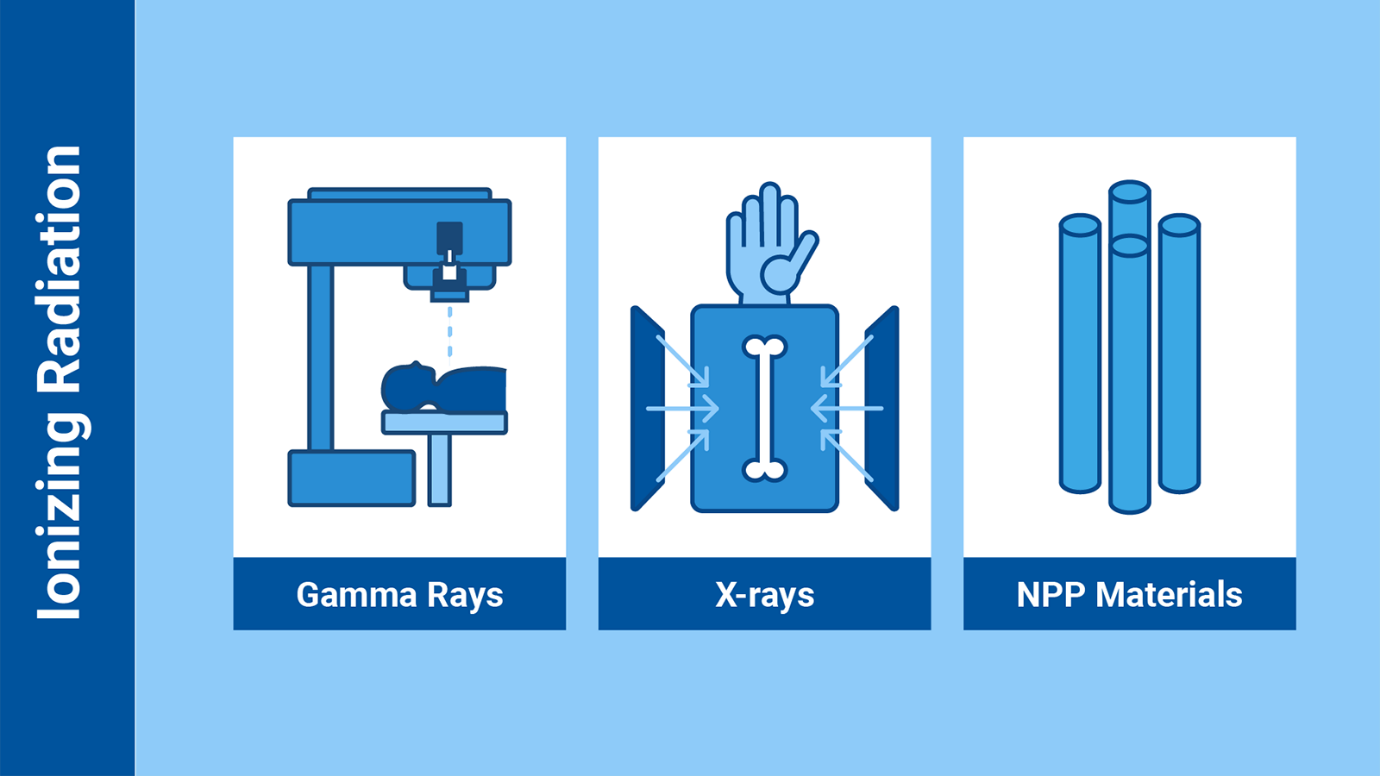
Some examples of ionizing radiation include some types of cancer treatments
using gamma rays, the X-rays, and the radiation emitted from radioactive
materials used in nuclear power plants (Infographic: Adriana Vargas/IAEA)
Ionizing radiation is a type of radiation of such energy that it can detach
electrons from atoms or molecules, which causes changes at the atomic level
when interacting with matter including living organisms. Such changes
usually involve the production of ions (electrically charged atoms or
molecules) – hence the term “ionizing” radiation.
For most people, non-ionizing radiation does not pose a risk to their health.
However, workers that are in regular contact with some sources of
non-ionizing radiation may need special measures to protect themselves from,
for example, the heat produced.
In high doses, ionizing radiation can damage cells or organs in our bodies or
even cause death. In the correct uses and doses and with the necessary
protective measures, this kind of radiation has many beneficial uses, such
as in energy production, in industry, in research and in medical diagnostics
and treatment of various diseases, such as cancer. While regulation of use
of sources of radiation and radiation protection are national
responsibility, the IAEA provides support to lawmakers and regulators
through a comprehensive system of international safety standards aiming to
protect workers and patients as well as members of the public and the
environment from the potential harmful effects of ionizing radiation.
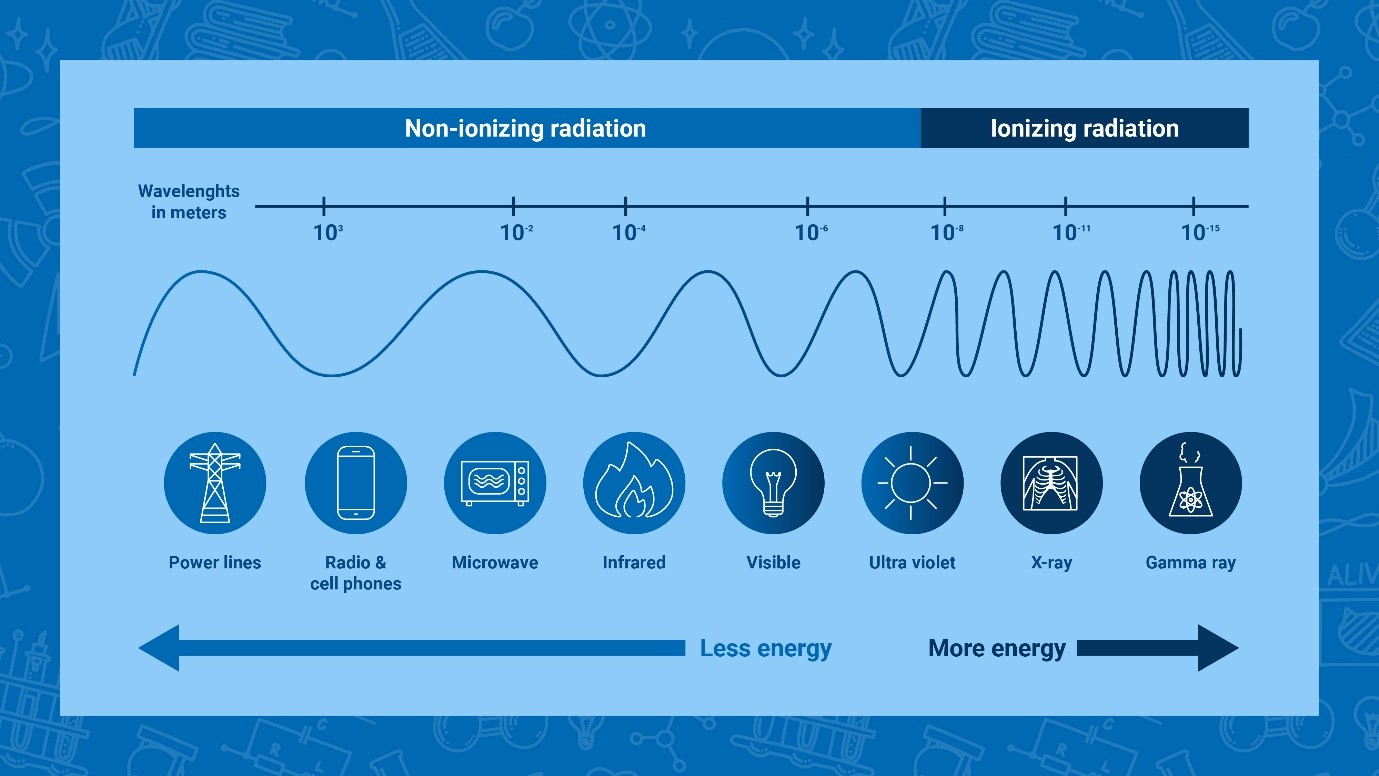
Non-ionizing and ionizing radiation have different wavelength, which directly
relate to its energy. (Infographic: Adriana Vargas/IAEA).
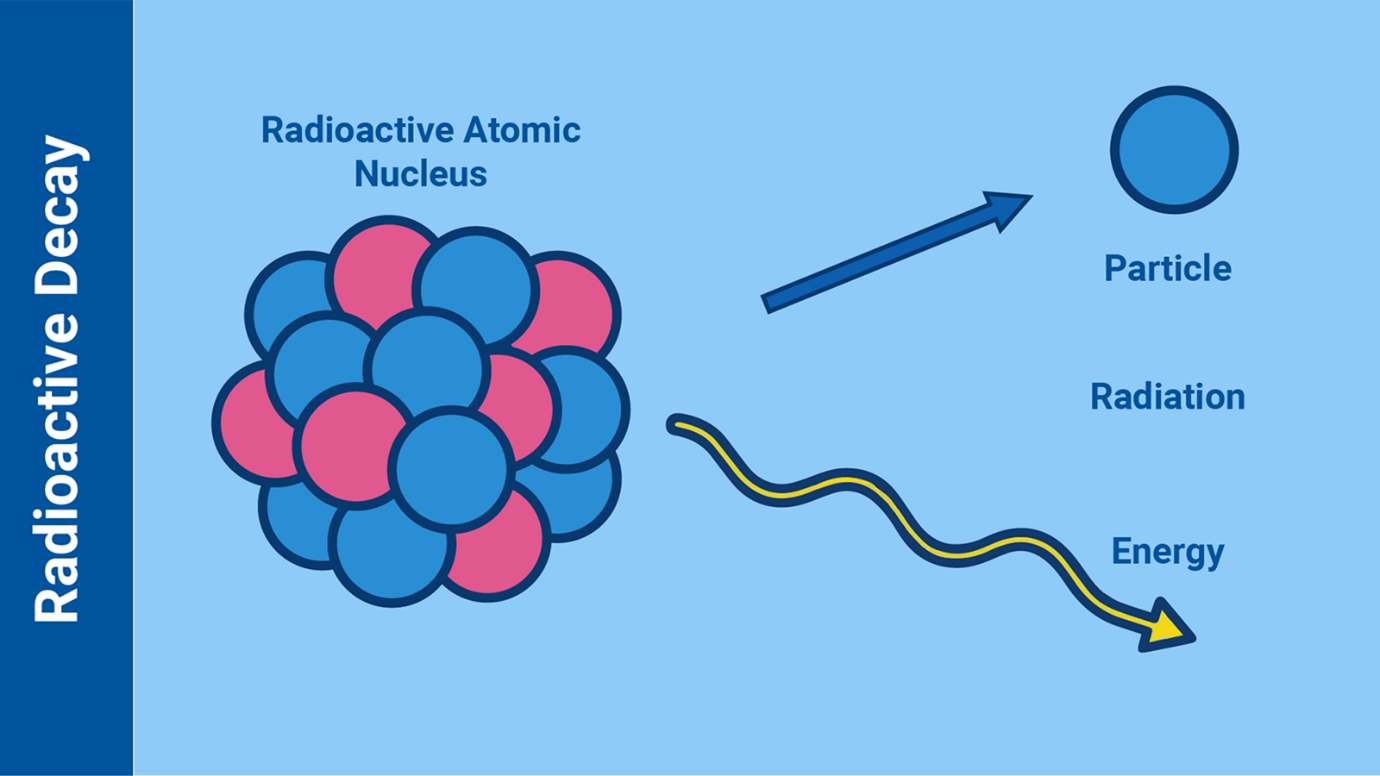
The science behind radioactive decay and the resulting radiation
Ionizing radiation can originate from, for example, unstable (radioactive)
atoms as they are transitioning into a more stable state while releasing
energy.
Most atoms on Earth are stable, mainly thanks to an equilibrated and stable
composition of particles (neutrons and protons) in their centre (or
nucleus). However, in some types of unstable atoms, the composition of the
number of protons and neutrons in their nucleus does not allow them to hold
those particles together. Such unstable atoms are called “radioactive
atoms”. When radioactive atoms decay, they release energy in the form of
ionizing radiation (for example alpha particles, beta particles, gamma rays
or neutrons), which, when safely harnessed and used, can produce various
benefits.
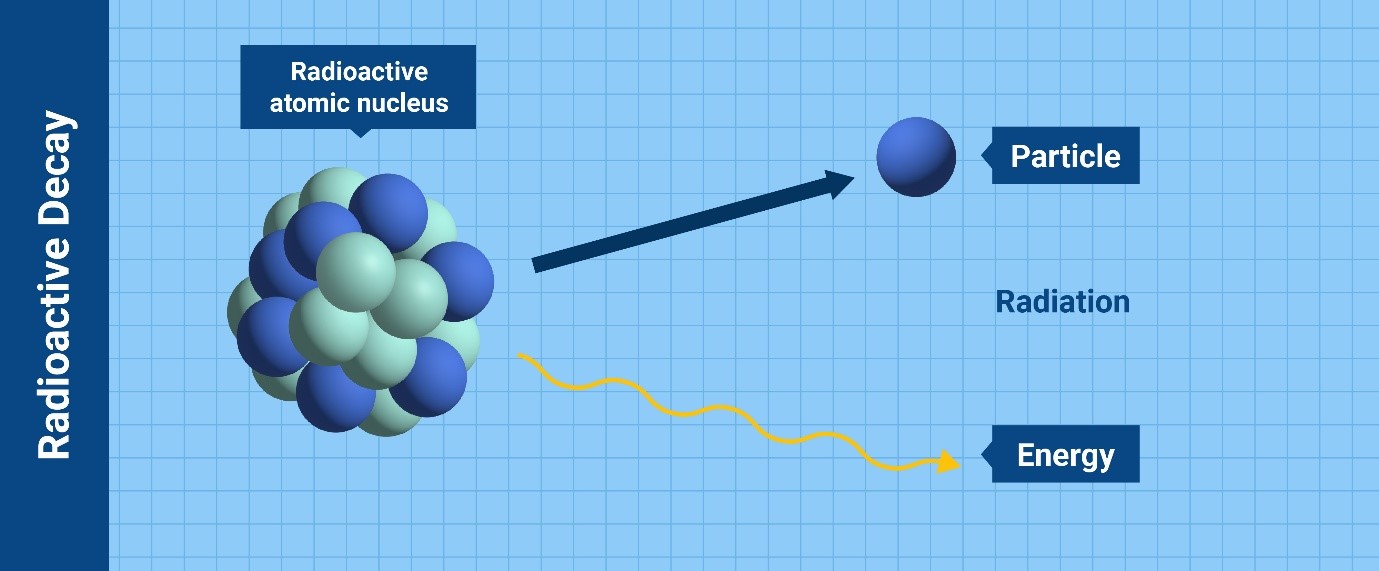
The process by which a radioactive atom becomes more stable by releasing
particles and energy is called “radioactive decay”. (Infographic: Adriana
Vargas/IAEA)
What are the most common types of radioactive decay? How can we protect
ourselves against the harmful effects of the resulting radiation?
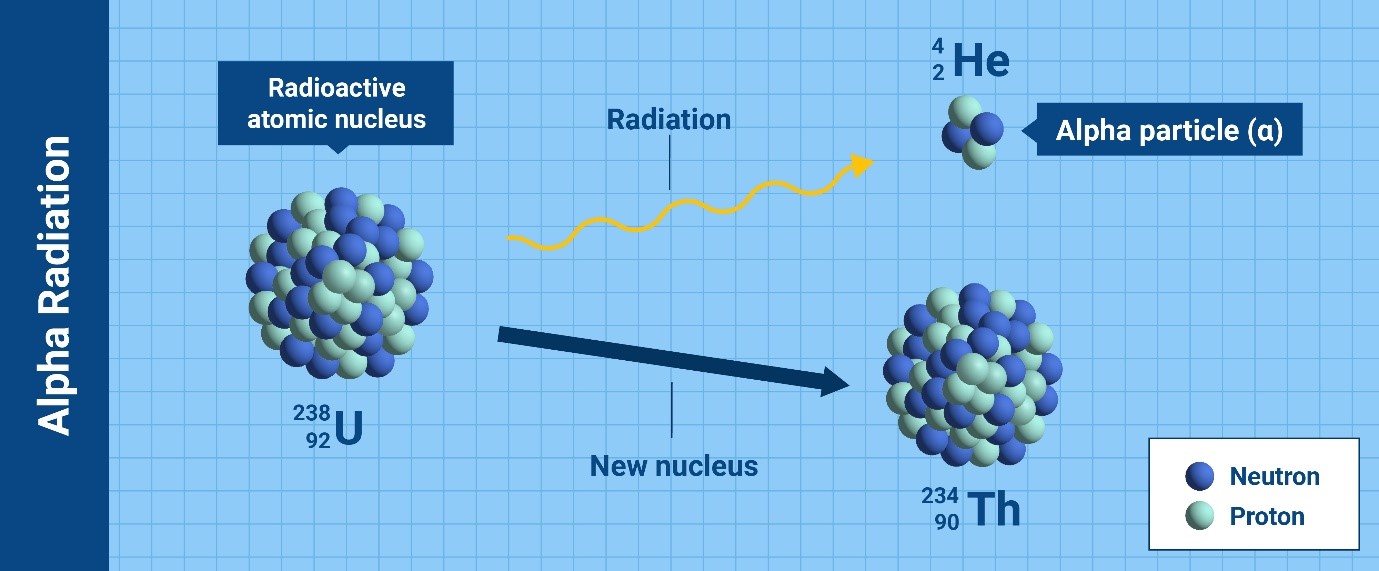
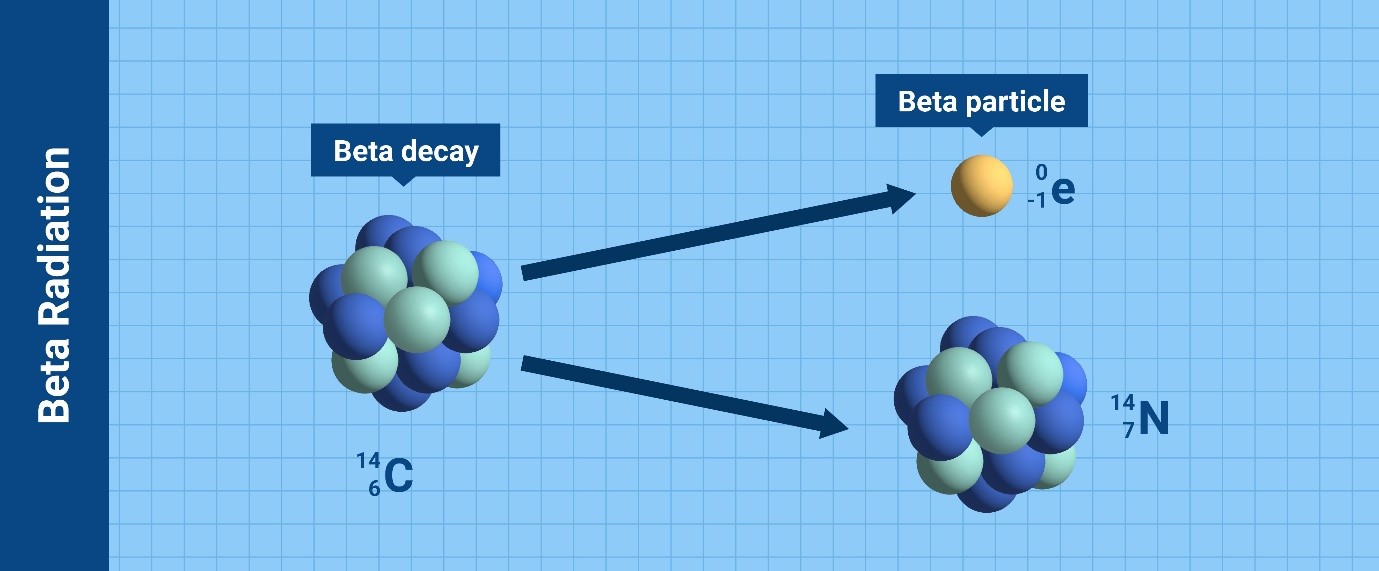
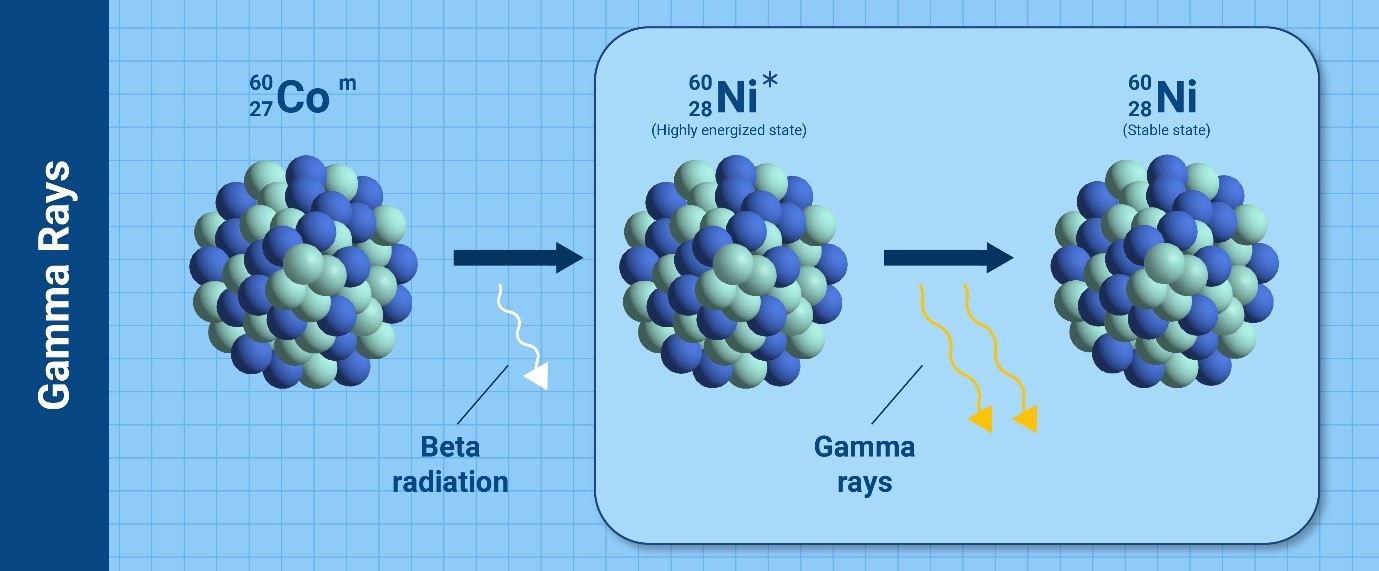
Depending on the type of particles or waves that the nucleus releases to
become stable, there are various kinds of radioactive decay leading to
ionizing radiation. The most common types are alpha particles, beta
particles, gamma rays and neutrons.
Radiation
Radiation is everywhere; living on this planet means being exposed to natural
radiation. Artificial radiation has been used successfully in the last
centuries for medical diagnosis and treatment of pathologies like cancer.
Not only the cosmos and our environment contain radioactivity. Even the
elements our bodies are made of are naturally found in different variants –
isotopes – some of which are radioactive, for example the radioisotopes of
potassium, caesium and radium.
Similar to visible light, radiation is of electromagnetic nature. When it is
powerful enough to break molecular bonds, thereby ionizing matter (the
process during which a neutral atom or molecule loses or gains electrons to
form ions), it is called ‘ionizing radiation.’ Molecular bonds may occur in
all materials, even in the building blocks of life – the DNA.
There is evidence that shows that alterations to DNA molecules caused by
ionizing radiation may generate mutated biological cells. The vast majority
of these mutations are not dangerous to human health, but there is a small
probability that some mutations may cause cancer. For this reason, it is
vital to understand how radiation interacts with biological matter.
Ionizing radiation can penetrate solid objects deeply. This characteristic is
the basis for diagnostic radiology and radiotherapy. X rays, one of the
forms of ionizing radiation, are emitted from an irradiation device on one
side of the object. The radiation that passes through the object is detected
by suitable detectors on the other side. This process can be used to produce
an image that shows the internal structures of the irradiated object without
opening it. When this process is applied in medicine, in a specialised field
called diagnostic radiology, it provides images of the internal structures
of the human body with minimal intervention.
In nuclear medicine, medical practitioners inject patients with a radioactive
substance that accumulates in a targeted part of the body. By detecting the
radiation exiting the body they can draw conclusions about the physiological
functions of the anatomy. In radiotherapy, radiation penetrates the body to
target and destroy tumours.
Natural sources make up around 80 per cent of the global average annual dose
people are exposed to. The largest artificial source of exposure for humans
is medical radiation. Its contribution to the total average annual dose is
around 20 per cent. This is about half of the contribution of the largest
natural component – radon inhalation in buildings – to the average annual
dose.
For this reason, it is important to minimize unwarranted medical exposures to
ionizing radiation. This is achieved by improving the processes of
justification and optimization of exposures. Justification requires that a
person may be exposed to radiation only when there is a clear net benefit
for him or her. Optimization processes on the other hand minimize the
radiation dose used to achieve a specific diagnostic or therapeutic result
to the lowest level that is achievable and reasonable.
What is an Atom?
Nuclear Explained
5 January 2026
Emma Midgley, IAEA Office of Public Information and Communication
An atom is the smallest unit of an element that retains its chemical
properties. It is made of protons, neutrons, and electrons. (Image: M.
Magnaye)
Atoms are the building blocks of matter. Everything around us — from air and
water, to rocks, plants and animals — as well as everything within our
bodies, is made up of atoms.
They are very small, the smallest units of an element that retain the
element’s chemical properties. The Ancient Greeks believed they were the
smallest particles in existence, and the word ‘atom’ is derived from
‘indivisible’ in Greek. A single strand of human hair is as thick as 500 000
carbon atoms stacked on top of each other.
This single atom of the metal strontium is visible in this photograph because
it has absorbed and re-emitted the light of a laser. The electrodes in the
picture are two millimetres apart. (Photo: David Nadlinger/Oxford
University)
Atoms cannot be seen with the naked eye, or even under a standard microscope.
An atom is too small to deflect visible light waves, meaning it will not
show up under light-focusing microscopes. Atoms can be viewed under an
electron microscope, which generate electron waves that can interact with
atoms. In the picture above, the atom is ‘visible’ because it has absorbed
and re-emitted the light of a laser.
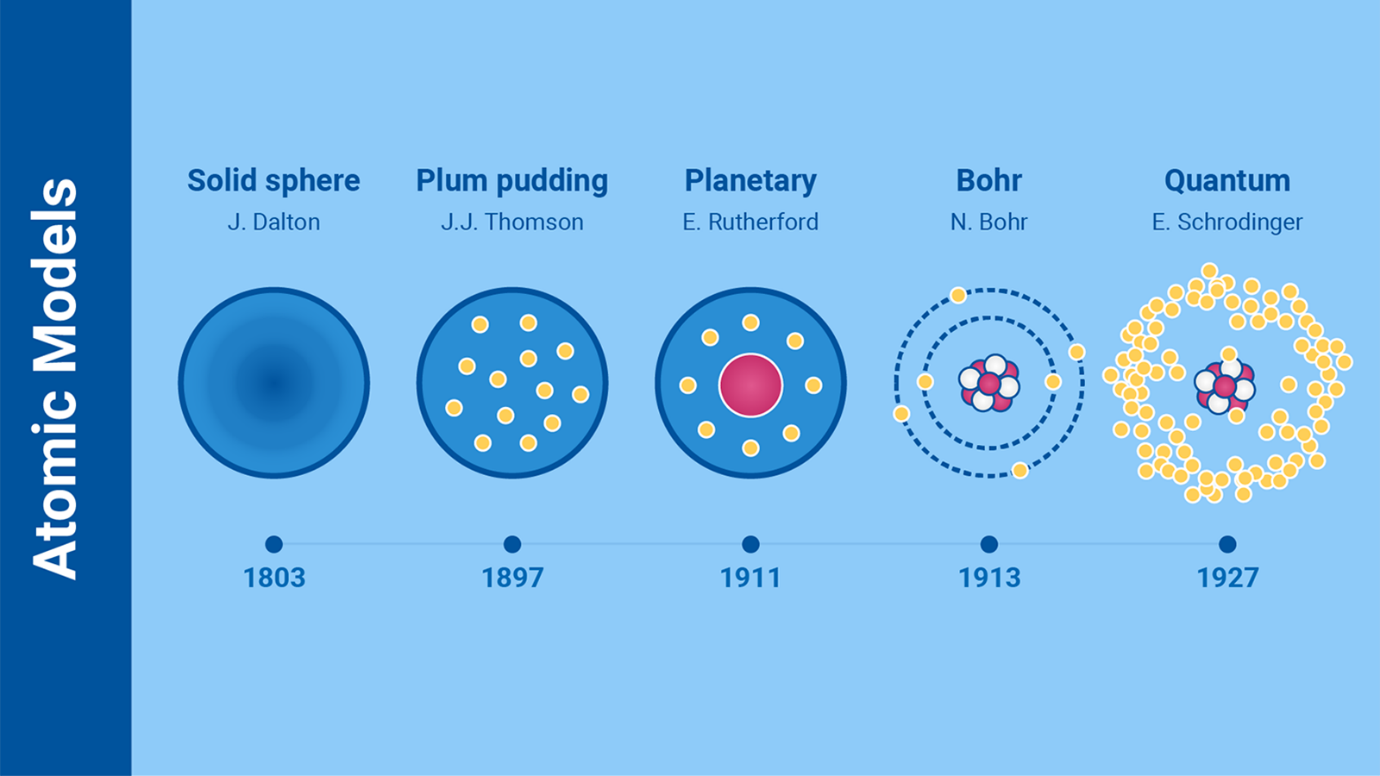
What do atoms look like? Scientists have changed their minds over the
centuries. (Infographic: M. Magnaye)
What are Atoms Made Of?
Each atom consists of three types of particles: protons, neutrons and
electrons. At the centre of an atom is a dense nucleus, which contains
protons and neutrons, and is much smaller than the entire atom. If the
nucleus of the atom were the size of a marble, the atom would be the size of
a sports stadium.
Protons have a positive electrical charge, while neutrons are neutral. The
nucleus stays together due to the ‘nuclear force’. This force binds the
protons and neutrons together at distances close to the size of the nucleus.
The nuclear force at this distance is much stronger than the electrical
repulsion between the protons (as they have equal charges, they would
otherwise repel each other). At larger distances this nuclear force rapidly
becomes insignificantly small.
The number of protons in an atom’s nucleus determines which element it is.
For example, an atom with one proton is hydrogen, while an atom with eight
protons is oxygen.
Surrounding the nucleus is a cloud of electrons — negatively charged
particles. The atomic nucleus and the electrons are bound together by
Coulomb force interactions – the forces in physics that describe the
repulsion or attraction between these charged particles. However, when an
electron gains energy, it can separate from the atom, causing the atom to
become a positively charged ion.
The atom at the centre of the IAEA’s logo has four electrons – meaning it is
Beryllium if it is neutral and not ionized. (Infographic: M. Magnaye)
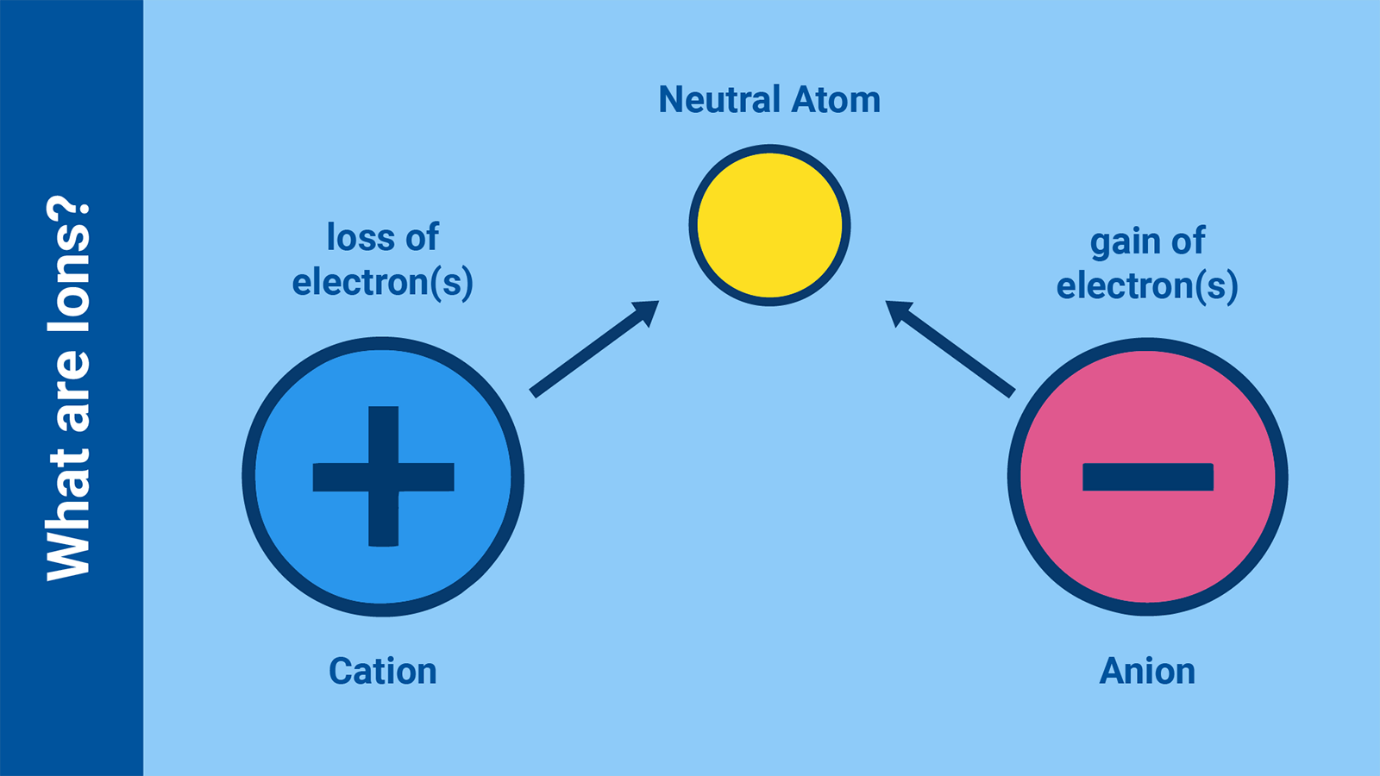
What are Ions?
Atoms with the same number of negatively charged electrons and positively
charged protons are neutral, as the charges cancel each other out. If an
atom gains or loses electrons it becomes an ion.
While the electric field of a neutral atom is weak, an electrically charged
or ionized atom has a strong electrical field, making it strongly attracted
to oppositely-charged ions and molecules. Atoms can be ionized by collisions
with other atoms, ions and subatomic particles. They can also be ionized by
exposure to gamma or X ray radiation. Ionizing radiation refers to radiation
that has enough energy to break an electron away from an atom. It can also
chemically alter material, for example damaging DNA in living tissue.
Most atoms on Earth are stable, mainly thanks to a balanced composition of
particles (neutrons and protons) in their nucleus.
However, in some types of unstable atoms, the composition of the number of
protons and neutrons in their nucleus does not allow them to hold those
particles together. In this case, the atom ‘decays’, and releases energy in
the form of radiation (for example alpha particles, beta particles, gamma
rays or neutrons), which, when safely harnessed and used, can produce
various benefits.
Read more: What are Isotopes?
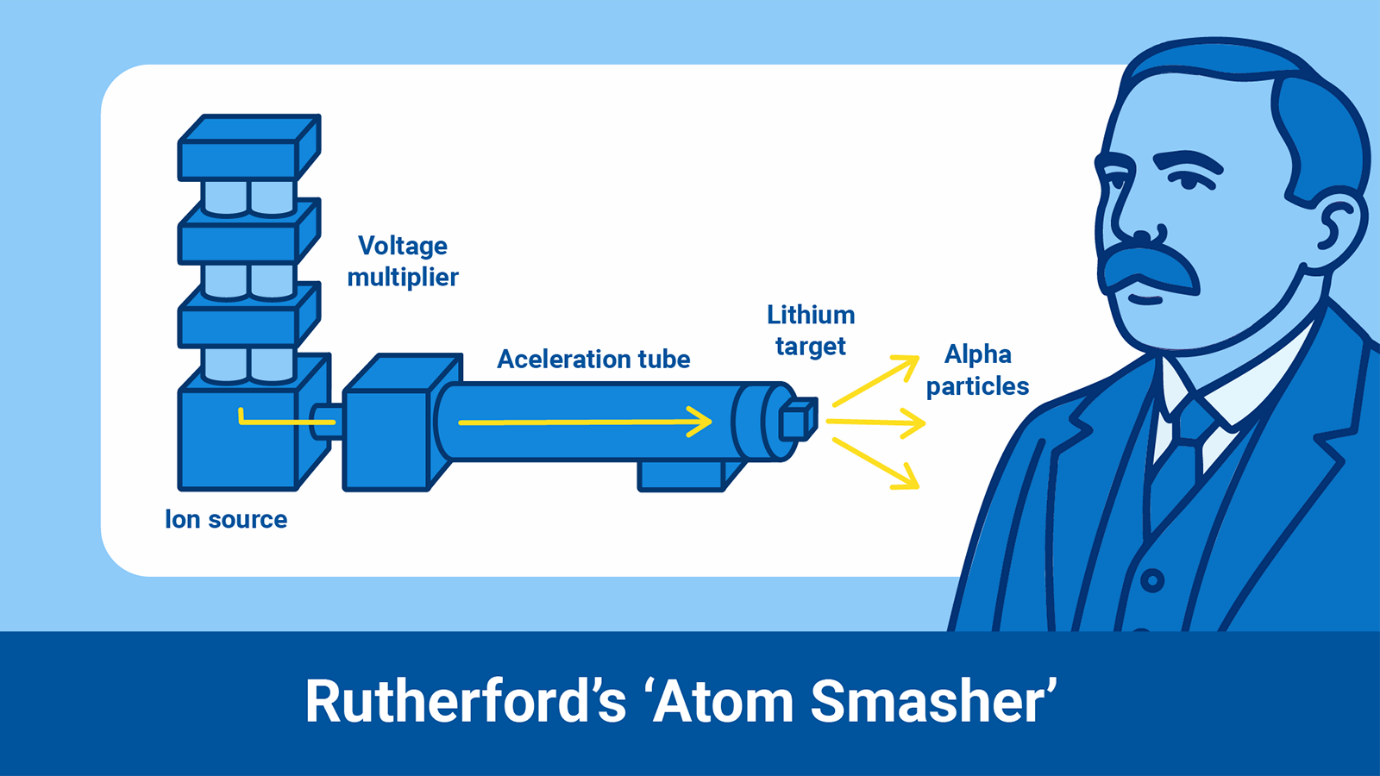
Ernest Rutherford: Inventor of the ‘Atom Smasher’
In 1917, a scientist called Ernest Rutherford discovered that by blasting
beams of radioactive alpha particles into nitrogen gas, the nitrogen atom
could be transmuted into oxygen while ejecting a hydrogen nucleus. This
subatomic particle (the hydrogen nucleus) was later renamed the proton.
Rutherford’s discovery led to the development of the first particle
accelerator, initially referred to as an ‘atom smasher’. This powerful
machine could accelerate charged particles using an electrical field to high
energies along a path and used strong magnets to create beams of single
charged particles. When the fast-moving particles hit the target (they could
go almost as fast as the speed of light), the atoms in the target split
apart.
Read more: What are particle accelerators?
Particle accelerators also can be used to create radioactive material by
shooting charged particles at atoms to change them into different, unstable
atoms, such as technetium-99m for medical imaging and radioisotopes for
targeted cancer therapy.
Today, particle accelerators are also used to sterilize medical equipment,
research the origins of the universe (for example, at the Large Hadron
Collider), as well as to analyse air samples and to enhance materials and
make them more resistant to damage. Different types of particle accelerators
include ion implanters, electron beam accelerators, cyclotrons,
synchrotrons, linear accelerators (Linacs) and electrostatic accelerators.
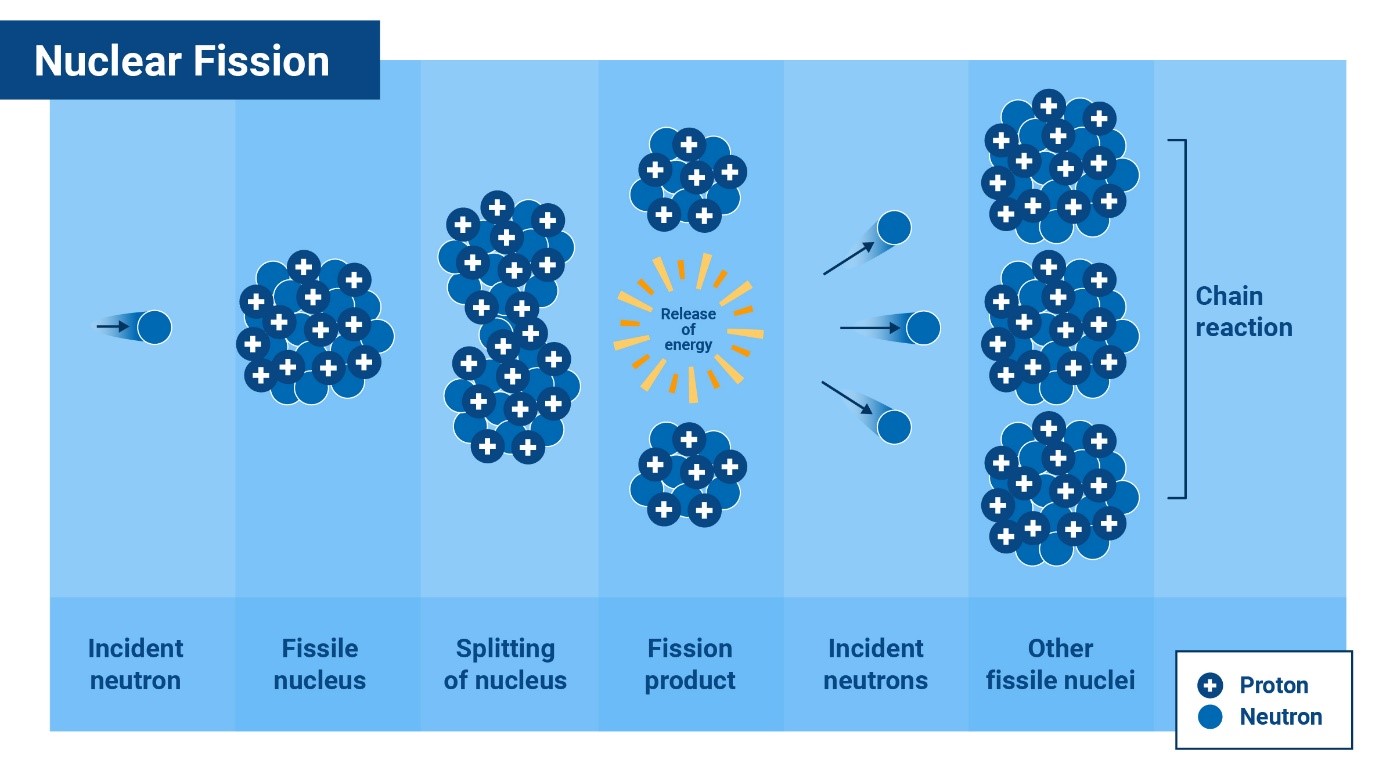
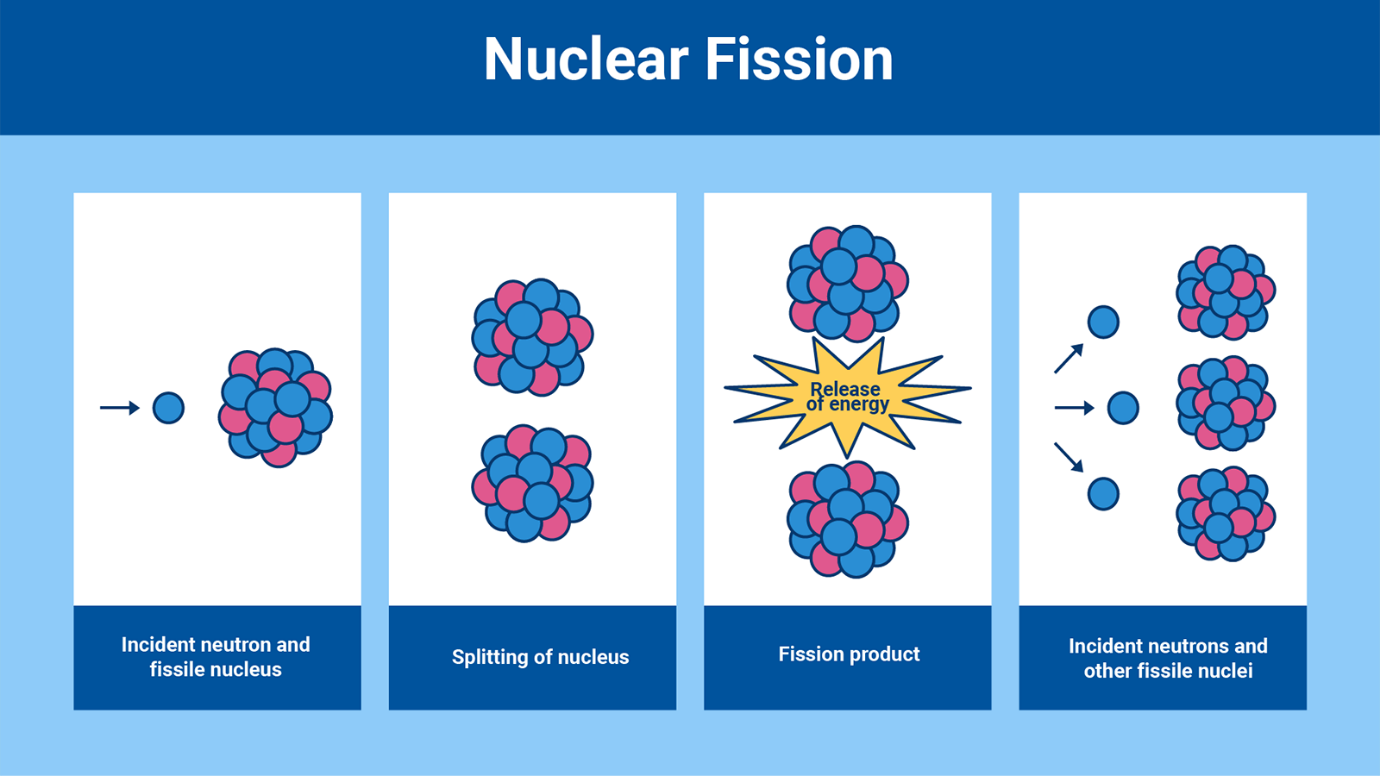
Splitting the Atom: Nuclear Fission
In the 1930s, scientists found out that if a neutron is fired into certain
uranium atoms, they could split into two and emit a certain number of
neutrons, releasing a huge amount of energy along the way. This is called
fission, from the Latin word for ’split’.
Uranium, with 92 protons, has the highest atomic number of all naturally
occurring elements on Earth. Uranium-235 is easier to split (fission) than
other isotopes because its nucleus is relatively unstable, and readily
absorbs a neutron, causing it to break apart into two lighter atoms.
However, only 0.7 per cent of uranium found on earth is this type of
uranium, described as fissile.
Read more about uranium here
Fission can be used to create a nuclear chain reaction. Every time a
uranium-235 atom is split it releases on average 2.5 neutrons. These can go
on to split further fissile nuclei, releasing yet more neutrons. However,
these ‘fast’ neutrons initially travel with too much energy to be effective
at causing fission. Using a ‘moderator’ such as water or graphite slows down
the neutrons. The neutrons lose most of their energy in collisions with the
hydrogen or carbon atoms to become ‘thermal’ or ‘slow’ neutrons which have a
much better chance of splitting other uranium nuclei.
The nuclear fission technique is now used to make 10% of the world’s
carbon-free energy — as nuclear fission produces no carbon dioxide.
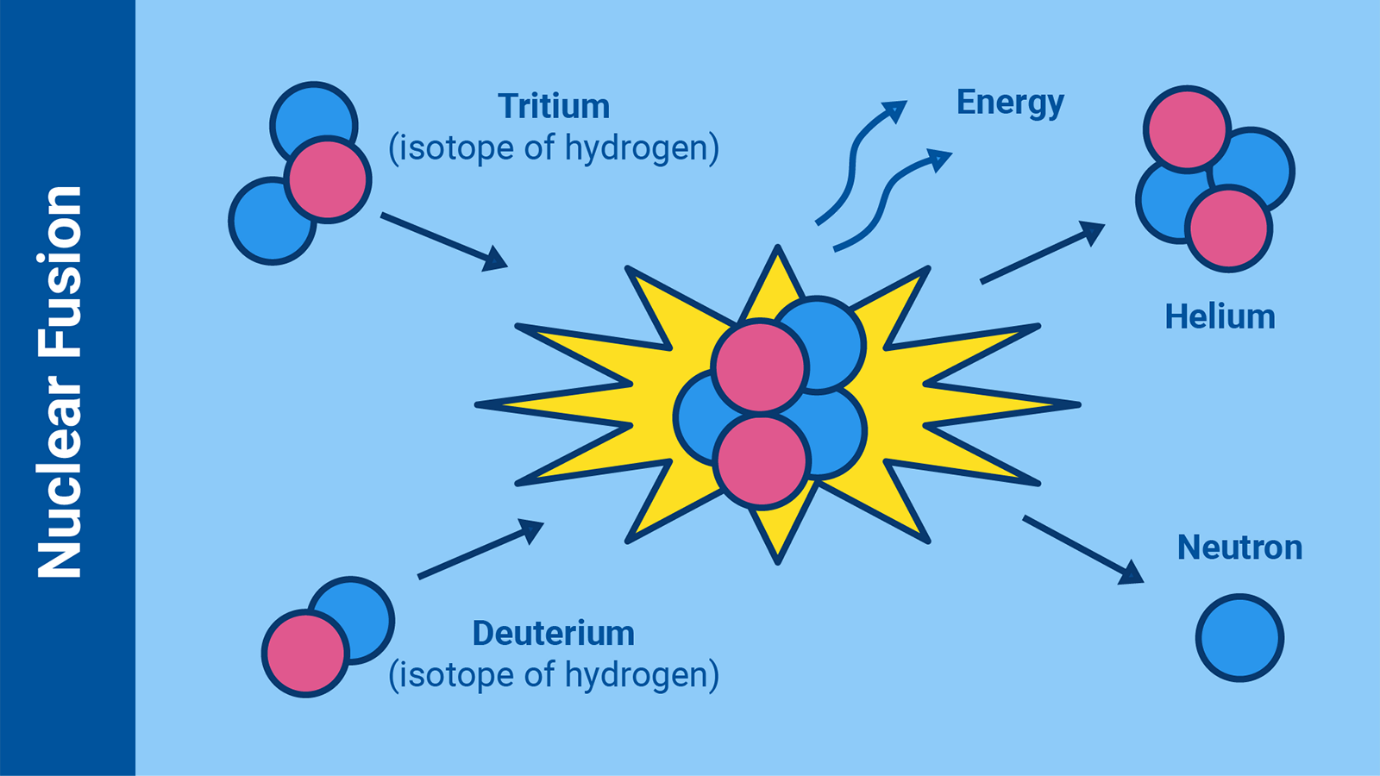
What happens to Atoms in Nuclear Fusion?
Nuclear fusion is the process by which two light atomic nuclei combine to
form a single heavier one while releasing massive amounts of energy, a
theory first understood in the 1920s.
Fusion reactions take place in a state of matter called plasma — a hot,
charged gas made of positive ions and free-moving electrons with unique
properties distinct from solids, liquids or gases.
The sun, along with all other stars, is powered by this reaction. To fuse,
nuclei need to collide with each other at extremely high temperatures,
around one hundred million degrees Celsius. The high temperature provides
them with enough energy to overcome their mutual electrical repulsion. Once
the nuclei come within a very close range of each other, the attractive
nuclear force between them will outweigh the electrical repulsion and allow
them to fuse. For this to happen, the nuclei must be confined within a small
space to increase the chances of collision. In the sun, the extreme pressure
produced by its immense gravity creates the conditions for fusion.
Radiation science examines the interaction of high-energy particles and
electromagnetic waves with matter, governed by processes such as ionization,
excitation, and nuclear transmutation. At the atomic level, ionizing
radiation displaces lattice atoms, producing vacancies, interstitials, and
defect clusters, which evolve under temperature and stress into macroscopic
material degradation.
Core Scientific Foundations
- Radiation-Matter Interaction:
Governed by cross-sections (σ), linear energy transfer (LET), and
stopping power.
- Displacement Damage: Measured
in displacements per atom (dpa), critical in reactor materials and
aerospace shielding
- Decay Kinetics: Exponential
decay models define isotope stability and half-life behavior.
Advanced Research Areas
- Radiation-resistant alloys (ODS steels, ceramic composites)
- High-flux neutron environments (fusion reactor simulation)
- Monte Carlo radiation transport simulations (MCNP, GEANT4)
- Real-time dosimetry using semiconductor detectors
Emerging Directions
- Self-healing materials that recombine defects under irradiation
- Nano-engineered shielding with enhanced attenuation efficiency
- Integration with AI for predictive radiation damage modeling
Key Challenges
- Long-term structural degradation in reactors
- Safe handling and disposal of radioactive waste
- Accurate modeling of mixed radiation fields